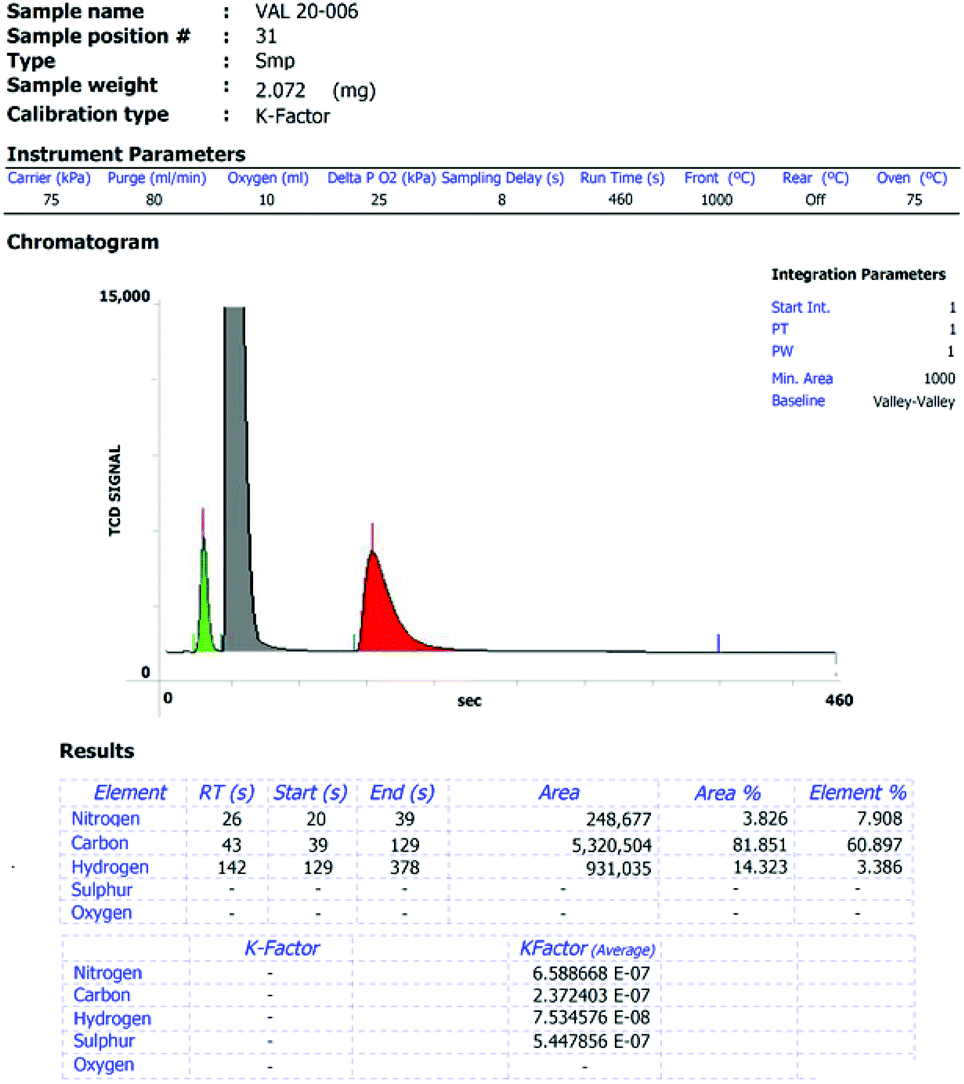
Elemental analysis: an important purity control but prone to manipulations - Inorganic Chemistry Frontiers (RSC Publishing) DOI:10.1039/D1QI01379C

Question Video: Determining the Impurity Percentage Given the Mass of the Desired Substance and the Total Mass | Nagwa

Elemental Impurities: Implications for Manufacturers of Drug Products, APIs, and Excipients | American Pharmaceutical Review - The Review of American Pharmaceutical Business & Technology