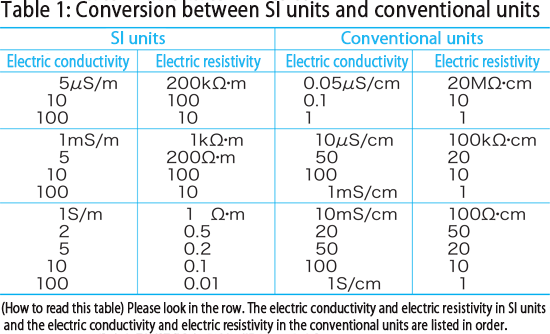
The conductivity of water at 298K is 0.55 × 10^ 7 S cm^ 1. if λ m(H^+)=350 S cm^2 mol^ 1 and λ(OH^ )=200 S cm^2, the degree of dissociation of water will b
At certain temperature the conductivity of pure water is 6.61/100000000ohm cm the ionic conductance of H+ and OH ions at this temperature are 350 and 200ohm 1 cm2 mol 1 respectively the














.webp)